|
CyanoWarm
|
|
Adaptive evolution to warming & acidification in potentially toxic cyanobacteria (CyanoWarm)
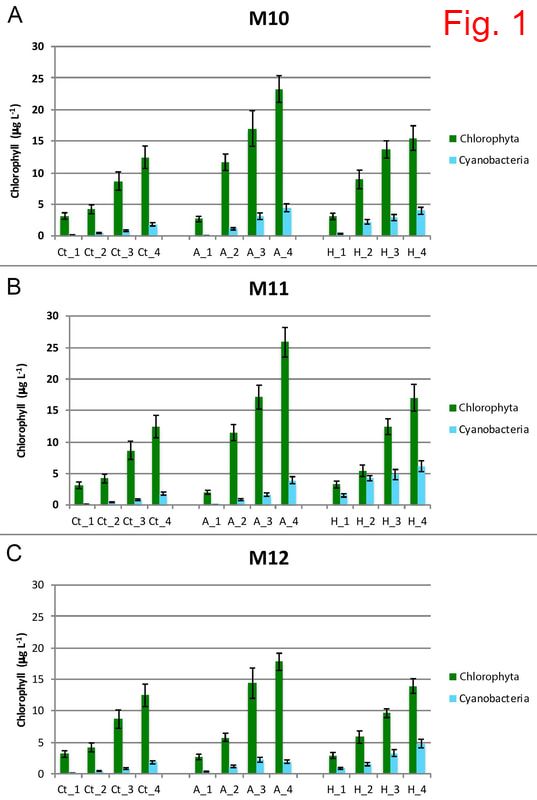
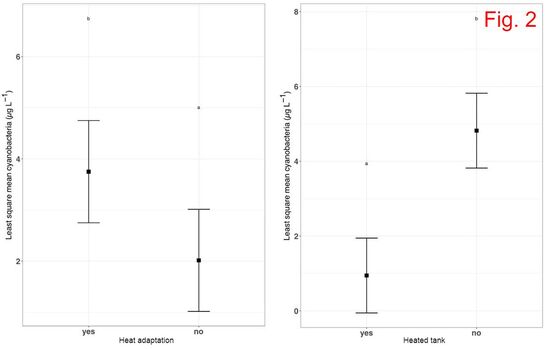
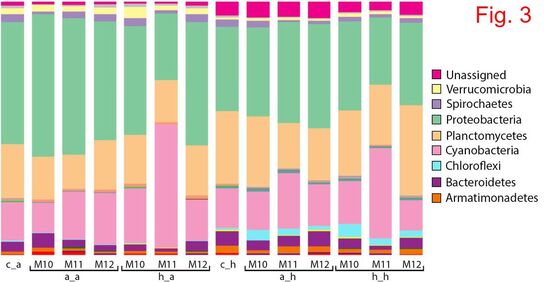
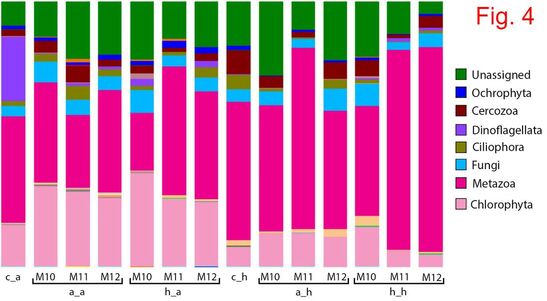
Climate change (including increase in water temperature and acidification – due to increased CO2) impacts the quality of the waters and the fundamental biology of our planet. This project aims to understand the evolutionary responses to warming and acidification in three key cyanobacterial genera: Microcystis, Anabaena and Planktothrix. Cyanobacteria are a globally distributed group of phytoplankton, producing a significant part of the atmospheric oxygen, and some species generate massive accumulations of biomass (blooms), which are often toxic. Knowing the response of cyanobacteria to climate change is crucial for understanding the long-term stability of aquatic ecosystems and occurrence of toxic blooms, and these aspects are still largely unknown. The predictions made by modeling techniques are mainly based on short-term experiments, but these experiments do not offer information on the evolutionary potential of these organisms. The specific aims of this project are: 1) to assess the adaptive potential of cyanobacteria to high temperature/acidification; 2) to investigate to what extent does pre-adaptation of multiple clones affect their evolutionary potential; 3) to check whether adaptation to increased temperature/acidification comes with any change in toxin production; 4) to understand to what extent the competitive ability of cyanobacteria is affected by/affects the natural communities of microorganisms. This project is funded by the Romanian Government (UEFISCDI), Project code: PN-III-P1-1.1 TE 2016-2110, Contract number: 72/2018. MAIN RESULTS (updated in December 2020) Growth rate evolution The growth rates of the cyanobacterial strains previously adapted for 12 months to 22°C and 26°C were measured in the control environment (22°C). At day 0, all growth rates were similar: μ=0.156-0.159. In some strains, a decrease in growth rate occurred after two months of exposure to both 22°C and 26°C, followed by a recovery after four months. In the case of the M. aeruginosa strain M11, there was no decrease in growth rate after two months. Moreover, growth rates of M11 continued to increase after four and six months, slightly faster for the strain adapted to 26°C. The growth rate of the strains after 12 months varied from µ=0.165-0.19, which translates in a doubling time of the cultures of approx. 0.40-0.43 day-1, meaning 150-160 generations. Competition experiments The experiments took place at the Research Station Seeon, LMU Munich, Germany. Each ambient/heat-adapted cyanobacterial strain strain was separately inoculated into 1.4L dialysis bags containing eutrophic lake water, which were immersed into plastic bags that were deployed into four 1000L water tanks. Community composition (in terms of main phytoplankton groups) was checked weekly (four weeks in total), using a AlgaeLab Analyzer. The most noteworthy results concernerd the abundance of chlorophytes and cyanobacteria (Fig. 1). The highest concentration of chlorophytes was measured in the dialysis bags inoculated with ambient-adapted M. aeruginosa cells. The abundance of cyanobacteria increased with time in all bags, but at a higher rate in those inoculated with the strains that were previously adapted to 26°C. Heat-adapted strain M11 reached the highest concentration after 4 weeks (6 µg L-1). Cyanobacteria, which occur naturally in most freshwater lakes from temperate regions, were also present in the control bags, but their concentration throughout the experiment was lower than in any of the communities inoculated with one of the three M. aeruginosa strains. ANOVA tests were conducted to check for differences between treatments at the time of the end sampling in the experiment (week 4). There were significant differences in cyanobacteria concentration among bags inoculated with the strains concerning the factor heat adaptation (except for strain M10) and the factor heated tank, but not concerning an interaction of the two factors (Fig. 2). Molecular tests - prokaryotes The most abundant prokaryotic OTUs identified in the samples belong to phylum Proteobacteria (26%-56% of total OTUs) (Fig. 3). Most of these OTUs were assigned to Alphaproteobacteria and Gammaproteobacteria. Cyanobacteria was the second well-represented phylum among the prokaryotic OTUs (12%-49%). By far, the highest abundance of cyanobacteria was found in the communities inoculated with heat-adapted M. aeruginosa strain M11, in both ambient (49% OTUs) and heated tanks (36% OTUs). Molecular tests - eukaryotes The eukaryotic populations in heated tanks were dominated by Metazoa (41%-77% of total reads), which were less abundant in the ambient tanks (22%-48% of the reads) (Fig. 4). Chlorophytes were identified in 16%-35% of the reads in the ambient tanks, and only in 5%-15% of the reads from the heated environments. Fungi (mostly belonging to group Cryptomycota) were identified in all samples (3%-9% of the reads). Dinoflagellates were found mostly in the communities inoculated with M. aeruginosa in the ambient tanks (1%-3%). MAIN CONCLUSIONS
|